RESEARCH
X-ray crystallography of protein
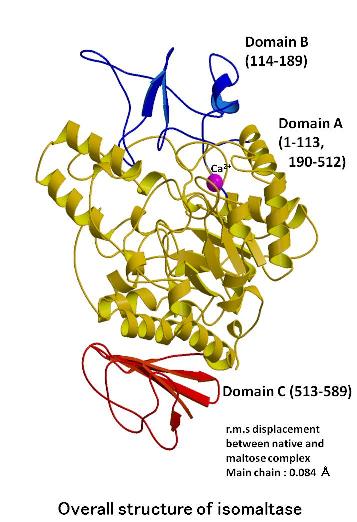
The structures of
isomaltase from Saccharomyces cerevisiae and in complex with maltose were determined at a resolution of 1.30 and
1.60 angstrom, respectively. Isomaltase contains 3 domains, namely, A,
B, and C. Domain A consists of the (b/a)8-barrel common to the glycoside hydrolase (GH) family 13. However, the
folding of domain C is rarely seen in other GH family 13 enzymes. An electron
density corresponding to a non-reducing end glucose residue was observed
in the active site of isomaltase in complex with maltose; however, only
incomplete density was observed for the reducing end. The active site pocket
contains 2 water chains. One water chain is a water path from the bottom
of the pocket to the surface of the protein and may act as a water drain
during substrate binding. The other water chain, which consists of 6 water
molecules, is located near the catalytic residues Glu277 and Asp352. These
water molecules may act as a reservoir that provides water for subsequent
hydrolytic events. The best substrate for oligo-1,6-glucosidase is isomaltotriose;
other longer chain oligosaccharides are also good substrates. However,
isomaltase shows the highest activity toward isomaltose and very little
activity toward longer oligosaccharides. This is because the entrance to
the active site pocket of isomaltose is severely narrowed by Tyr158, His280,
and loop 310-315, and because isomaltase pocket is shallower than that
of other oligo-1,6-glucosidases. These features of the isomaltase active
site pocket prevent isomaltooligosaccharides from binding to the active
site effectively.
Banners
SAKAI Laboratory
Postal Code 634-0813
Shijo-cho 88, Kashihara,
Nara, Japan